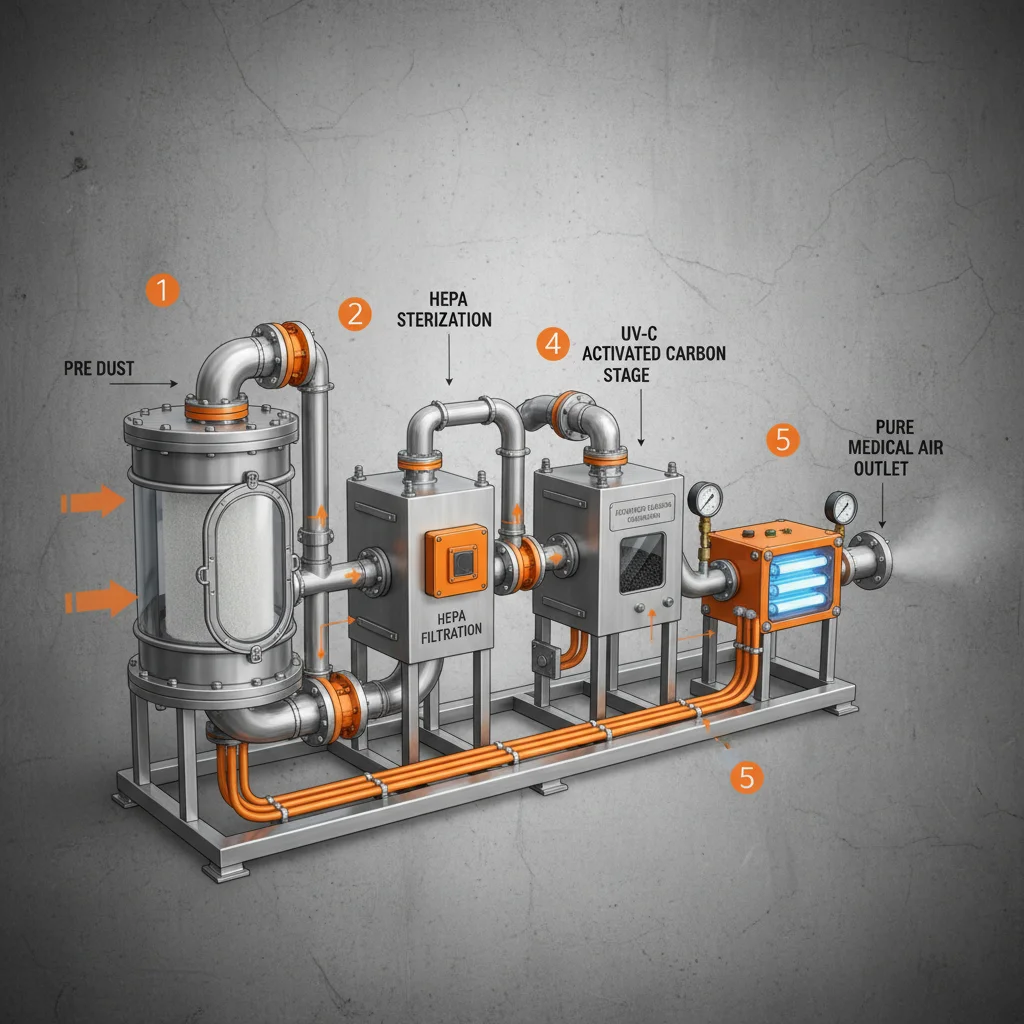
The global demand for sterile environments has surged, positioning the air disinfection machine factory as a cornerstone of modern public health infrastructure. By integrating advanced filtration and ultraviolet sterilization, these facilities ensure that the air we breathe in hospitals, laboratories, and homes is free from harmful pathogens.
Understanding the operational excellence of an air disinfection machine factory is crucial for procurement officers and health administrators who require reliable, high-capacity equipment. These factories do not merely assemble parts; they engineer complex systems that balance airflow efficiency with maximum microbial kill rates, adhering to stringent international safety standards.
From the perspective of industrial manufacturing, the synergy between high-grade silicone sealing components and electronic sterilization modules defines the quality of the final product. This comprehensive guide explores how a leading air disinfection machine factory optimizes its production to deliver sustainable, efficient, and life-saving air purification solutions worldwide.


In an era marked by emerging viral threats and increasing urban pollution, the role of the air disinfection machine factory has transitioned from a niche industrial provider to a critical pillar of global health security. According to ISO air quality standards, the ability to maintain "clean room" environments is no longer limited to pharmaceutical labs but is now a requirement for public transportation hubs and office complexes.
The challenge lies in the rapid deployment of technology that can handle high volumes of air without compromising energy efficiency. Factories are now utilizing AI-driven assembly lines to ensure that every unit produced meets rigorous efficacy benchmarks, reducing the risk of cross-contamination in high-traffic public areas.
At its core, an air disinfection machine factory is a specialized manufacturing facility dedicated to the design, fabrication, and testing of devices that eliminate airborne microorganisms. These factories integrate mechanical engineering with microbiology to create systems capable of neutralizing bacteria, viruses, and mold spores using methods such as UVC radiation, HEPA filtration, or plasma ionization.
Beyond simple assembly, these factories serve as innovation hubs where materials science is applied to improve the longevity of the machines. For instance, the use of medical-grade silicone for sealing rings ensures that sterilized air does not leak and that contaminated air is not drawn back into the clean zone, highlighting the intersection of rubber manufacturing and health technology.
The connection to humanitarian needs is profound; in regions with underdeveloped healthcare systems, a reliable air disinfection machine factory provides the necessary equipment to turn standard rooms into temporary surgical theaters, thereby saving countless lives during medical emergencies.
Durability is the primary focus within a professional air disinfection machine factory, as these devices often run 24/7 in critical environments. The structural integrity of the chassis and the quality of the internal seals are paramount to prevent systemic failure.
Scalability allows an air disinfection machine factory to pivot between producing small residential units and massive industrial HVAC integrations. This flexibility is achieved through modular design, where the same core sterilization engine can be adapted for different flow rates and room volumes.
Cost efficiency is managed not by cutting corners, but by optimizing the supply chain. By sourcing high-quality components, such as precision-molded silicone gaskets, an air disinfection machine factory can reduce maintenance intervals and lower the total cost of ownership for the end-user.
Measuring the success of production in an air disinfection machine factory requires a data-driven approach. Key performance indicators (KPIs) typically include the "Clean Air Delivery Rate" (CADR) and the "Single-pass Kill Rate," which determine how effectively a machine can sanitize a given cubic meter of air.
To ensure consistency, factories implement rigorous stress tests on their components. By comparing different sterilization technologies, manufacturers can determine which method provides the best balance of safety and speed for specific industrial applications.
The outputs of a high-capacity air disinfection machine factory are deployed across diverse sectors. In healthcare, they are used in operating theaters to prevent surgical site infections, while in the food industry, they protect processed goods from airborne contaminants during packaging.
In remote industrial zones or post-disaster relief operations, portable units from an air disinfection machine factory provide a safe haven for medical staff and displaced populations, ensuring that respiratory illnesses do not spread in crowded temporary shelters.
Investing in a partnership with a reputable air disinfection machine factory yields tangible long-term benefits, primarily through the reduction of sick-leave and the enhancement of occupational safety. By creating a biologically secure environment, companies can ensure the continuity of their operations even during seasonal flu outbreaks.
From a sustainability angle, modern factories are focusing on "green sterilization." This involves developing lamps with longer lifespans and filters made from biodegradable or recyclable materials, reducing the environmental footprint of air purification.
Ultimately, the value lies in the trust and dignity provided to the end-user. Knowing that the air is being managed by a system engineered in a world-class air disinfection machine factory allows employees and patients to feel secure and valued.
The next generation of air disinfection machine factory production will be driven by the "Internet of Medical Things" (IoMT). We are seeing the rise of smart sensors that detect pathogen loads in real-time, automatically adjusting the sterilization intensity to save energy when the room is unoccupied.
Material science is also evolving, with the introduction of nano-coatings that prevent biofilms from forming on the interior surfaces of the machines. This reduces the need for chemical cleaning and extends the operational life of the equipment.
Automation in the factory itself is increasing, with robotic assembly ensuring micron-level precision in the placement of sealing rings and filters, which is critical for maintaining the airtight integrity of high-pressure disinfection systems.
| Technology Type | Primary Application | Maintenance Level | Energy Efficiency |
|---|---|---|---|
| UVC Radiation | Hospital Wards | Medium (Lamp Replace) | High |
| HEPA H14 | Clean Rooms | High (Filter Change) | Medium |
| Cold Plasma | Industrial Plants | Low | Very High |
| Photocatalytic | Office Spaces | Low | High |
| Electrostatic | Public Transit | Medium | Medium |
| Hybrid Systems | Pharmaceuticals | High | Low |
A professional factory implements "fail-safe" mechanisms, including motion sensors and shielded chambers, to ensure that UVC light never contacts human skin or eyes. Rigorous leak testing is performed on all housing units to prevent radiation leakage.
With regular maintenance, industrial-grade units typically last 5-10 years. The lifespan depends heavily on the quality of the internal components, such as the silicone seals and motor bearings, which prevent wear and tear.
Yes, many factories specialize in "in-duct" disinfection modules. These are designed to be installed directly into existing ventilation shafts, allowing for the sterilization of air as it circulates through a building without requiring new standalone units.
Look for ISO 9001 (Quality Management) and ISO 14001 (Environmental Management) certifications. Additionally, check for CE or UL markings on the products, which indicate that the factory adheres to international safety and electrical standards.
Leading factories are transitioning to washable, permanent filters or those made from recycled polymers. Some are even experimenting with biological filters that naturally break down pollutants, reducing landfill waste.
Purification typically refers to the removal of particles (dust, pollen) via filters. Disinfection, as provided by an air disinfection machine factory, involves the actual killing or neutralizing of biological pathogens like viruses and bacteria.
The sophistication of a modern air disinfection machine factory represents the critical intersection of industrial manufacturing and public health. By focusing on durability, scalability, and the integration of high-performance materials like medical-grade silicone, these factories provide the essential tools needed to combat airborne threats and maintain sterile environments across the globe.
As we look toward the future, the shift toward smart, automated, and sustainable sterilization will further redefine how we interact with our indoor environments. For organizations seeking to enhance their air quality and ensure the safety of their occupants, partnering with a certified, innovation-driven factory is the most effective strategy for long-term resilience. Visit our website: www.gyfsilicone.com